
Biotech companies don’t scale on headcount—they scale on retained intelligence. Every assay optimization, regulatory workaround, vendor nuance, and experimental failure encodes knowledge that compounds enterprise value. When that knowledge lives in people instead of systems, your company is fragile by default.
In early biotech, speed rewards intuition and heroics. But past ~30–40 FTEs, intuition becomes a bottleneck. Knowledge fragments across Slack, notebooks, lab benches, and one “go-to” scientist. The risk isn’t just inefficiency—it’s irreversibility. Once knowledge leaves, you don’t get it back. You re-learn it at 10x the cost, under deadline pressure, often with regulators watching.
Biotech knowledge loss prevention is not documentation theater. It is a strategic discipline: converting tacit know-how into institutional memory that survives attrition, acquisitions, audits, and scale.
The Biotech Risk (What Happens If You Ignore This)
Most biotech operators underestimate knowledge loss because it doesn’t show up on the P&L—until it detonates.
Here’s the pattern:
A senior scientist leaves. Suddenly, experiments stall.
A regulatory lead departs. Submissions slow, questions pile up.
A manufacturing SME exits. Yield drops, timelines slip.
The hidden cost?
Lost capital: Delays extend burn by months. Investors notice execution drag.
Lost trust: Boards and partners sense fragility when progress depends on individuals.
Lost time: Teams re-discover decisions already made—often incorrectly.
In diligence, acquirers and late-stage investors probe operational continuity. If knowledge is person-bound, valuation discounts follow. In extreme cases, programs are paused because no one can explain why things were done a certain way.
Knowledge loss doesn’t feel dramatic. It feels like “we’re slower than we should be.” That’s why it’s so dangerous.
Framework: The Biotech Knowledge Continuity System
This system has four layers, applied sequentially.
1. Identify Mission-Critical Knowledge
Not all knowledge is equal. Focus on:
Regulatory rationale (why decisions were made)
Experimental design logic (not just protocols)
Vendor and CRO edge cases
Manufacturing and scale-up learnings
Rule: If losing this knowledge delays milestones by >30 days, it’s critical.
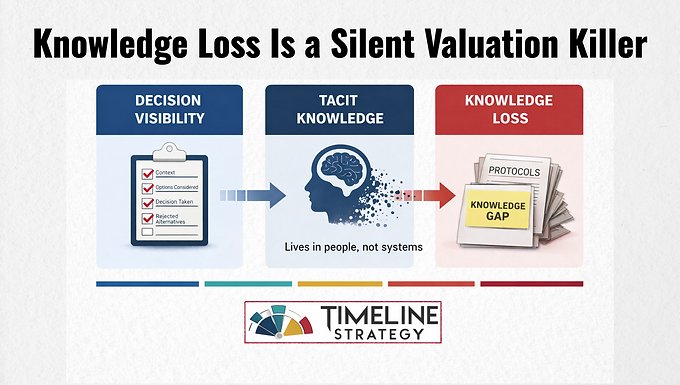
2. Convert Tacit → Explicit
Tacit knowledge lives in heads. Explicit knowledge lives in systems.
Use Decision Logs, not essays:
Context
Options considered
Decision taken
Why alternatives were rejected
This mirrors FDA and investor thinking—and prevents re-litigation.
3. Embed Knowledge Into Workflow
Documentation that lives “somewhere” dies.
Embed knowledge into:
SOPs linked to rationale
Experiment templates with decision history
Onboarding checklists by function
If it’s not in the flow of work, it won’t be used.
4. Assign Knowledge Ownership
Every critical knowledge domain needs:
A primary owner
A secondary shadow
A review cadence (quarterly minimum)
Ownership turns knowledge from an artifact into an asset.
Diagnostic Exercise (CEO / CSO Quick Test)
Answer these honestly:
If your top 3 scientists left tomorrow, what breaks first?
Can a new hire explain why your lead program is designed the way it is?
Where are regulatory decisions documented—email, memory, or system?
Who owns knowledge continuity in your org today?
Score yourself:
0–1 “clear answers” → High fragility
2–3 → Moderate risk
4 → Scalable foundation
If you hesitate, knowledge loss is already happening.
Insider Tip (Advanced Insight)
The highest ROI move is not documentation—it’s exit simulations. Once per quarter, ask: “If X left, what would we lose?” Then capture only that. This focuses effort where valuation risk actually lives and avoids bloated knowledge bases no one reads.
Closing
Want to harden your biotech against knowledge loss and de-risk scale?
→ Book a Strategy Workshop and we’ll map your critical knowledge assets in 90 minutes.
